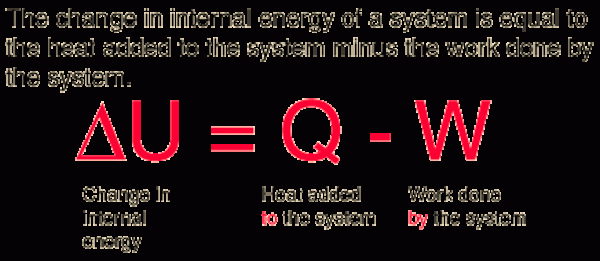

Thermodynamics is a branch of physics concerned with heat and temperature and their relation to energy and work. It defines macroscopic variables, such as internal energy, entropy, and pressure, that partly describe a body of matter or radiation.
Kinetic Energy = KE
= 1/2 mv2
Vebuity V = 0
mometntump = mv = 0
Heat is energy ------------------- energy = Joule
Heatis proportional of the KE of the modeculas of the suvstance.
Temperature is a number. If it exprssed in veluinon degree contorgoade.
Temperature is related of the average KE(Kelvin) of the molecles of the substance.
Thermocouple
_LMB.png) The Voltage can be interpreted to measure temperature.
The Voltage can be interpreted to measure temperature.
Zeroth law of thermolynamics
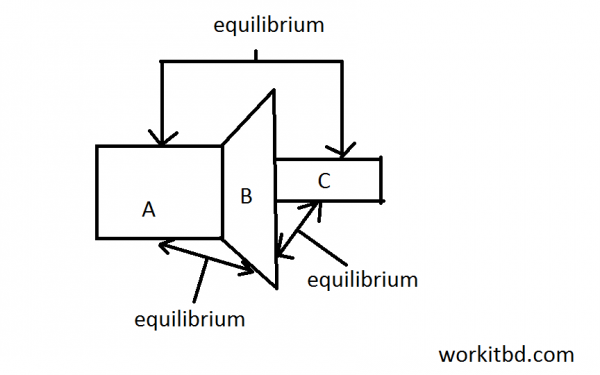
This is states zeroth law of Thermodynamics
First Law of Thermodynamics:
The first law of thermodynamics is the application of the conservation of the energy principle to heat and thermodynamic process.
The change in the internal energy of a system is equal to the heat added to the system minus the work done by the system.
The change in the internal energy of a system is equal to the heat added to the system minus the work done by the system.
first underline is for '<|U'
secound underline is for 'Q'
(-) for minus and last underline is for 'W'
<|U=Q-W
<|U = Change in internal system
Q = Heat added to the system
W = Work done by the system
Internal Energy
Internal energy is related to the kinetic energy and potential energy of the molecules of the substance.
W= pdv
Specific Heat
The specific heat is the amount of heat per unit mass required to raise the temperature by one degree celcias.
The relation between heat and temperature is usually expressed in the form shown below.
C=Q/m<|T , Were 'C' is the specific heat
Unit of C is = joule/gmoc
Kainatic Enargy:-
KE = 1/2mv2
v = velocity
The relation does not apply if a phase change is encounterel, because the heat does not change the temperature.
Temperature is related to the average kinatic enargy.
1/1= infinati , Q/infinati = 0
C = Q/m<|T
C = Q/m x 0, where '<|T = 0'
C = Q/0
C = Infinati
Constant Volume specific Heat
Using 1st law of thermodynamics
<|U = Q - W
= Q [w=pdv=p<|v=0]
= Cvn<|T
Cv = <|U/n<|T .............................(I)
Cv = constant volume spacific hit
for monoatonic ideal gas:-
U = 3/2 nrT
<|U = 3/2 nr<|T .................. (2)
from (1) and (2)
Cv = 1/n<|T 3/2 nR<|T
Cv = 3/2 R
'r' = Monoatomic gas.
Constant Pressure Specific Heat
Cp = Cv + R [Have to remember]
Cp = Cv + R
= 3/2 R + R
= 3/2 R + 2/2 R
= 5/2 R